The Science Behind Copper Peptide Analysis
In the expanding field of bioinorganic chemistry, Copper peptide analysis has become a defining tool for understanding metal–ligand interactions at the molecular level. Among the most extensively studied complexes, GHK-Cu — composed of glycine, histidine, and lysine bound to copper(II) ions — serves as a gold-standard model for investigating metal coordination, ligand geometry, and redox stability.
Laboratories worldwide use GHK-Cu as a representative system for exploring peptide–metal behavior under controlled, reproducible conditions. Because of its well-defined structure, it allows scientists to test hypotheses about copper-binding kinetics, metal exchange rates, and spectroscopic fingerprints.
Modern analytical chemistry now leverages advanced spectroscopic and chromatographic techniques to characterize this peptide precisely. These methods reveal not only how copper binds but why the resulting coordination geometry stabilizes so efficiently.
This guide presents a detailed exploration of analytical approaches to GHK-Cu characterization, highlighting best practices, integrated workflows, and emerging trends that continue to push the boundaries of peptide-metal research.
For those interested in learning more, accessing accurate and updated resources can help guide smarter decisions—this information on Peptides Costa Rica serves as a helpful starting point for exploring available options and understanding the broader landscape of peptide use in the region.
Fundamentals of Copper Peptide Analysis
Understanding Metal Coordination
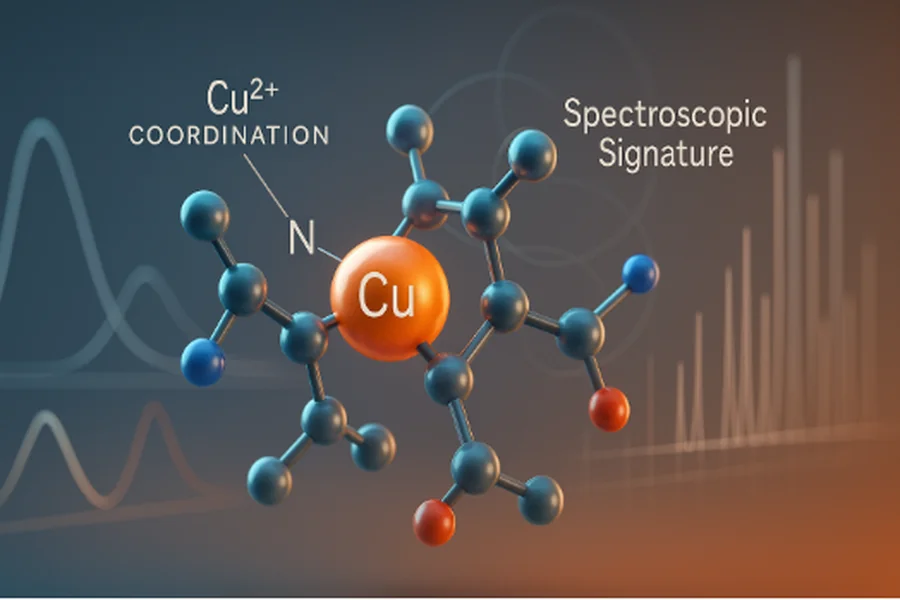
In its most stable form, GHK-Cu forms a square-planar complex in which the copper ion coordinates through nitrogen donors from histidine and the amine groups of glycine and lysine. Oxygen atoms from carboxyl groups complete the chelation. This arrangement provides a balance between flexibility and stability, creating a robust coordination framework.
The chelation environment determines how effectively GHK binds or releases copper under laboratory conditions. The study of this relationship forms the cornerstone of Copper peptide analysis.
The Significance of Copper-Binding Kinetics
Copper-binding kinetics define how fast the copper ion associates with or dissociates from GHK. These kinetics depend on solution pH, ionic strength, temperature, and competing ligands. Understanding rate constants allows researchers to predict complex behavior during analytical runs.
Kinetic data typically come from time-resolved spectroscopy, which records absorbance changes as the copper ion binds. Faster binding rates indicate higher affinity, while slower release suggests strong chelation.
Such kinetic studies inform how to design experiments for precise and reproducible results.
Key Takeaway
The structural geometry of GHK-Cu directly determines its copper-binding rate and stability under analytical conditions.
Spectroscopic Techniques in Copper Peptide Analysis
Spectroscopy provides the fastest and most detailed insight into molecular coordination and metal-ligand interactions. Each technique reveals complementary data, contributing to a holistic understanding of GHK-Cu.
Ultraviolet–Visible (UV–Vis) Spectroscopy
UV–Vis spectroscopy remains the foundation for GHK-Cu studies. When GHK binds copper, ligand-to-metal charge transfer transitions occur, resulting in distinct absorption peaks around 600–800 nm.
Researchers conduct titrations by adding incremental copper concentrations to GHK solutions and recording absorbance shifts. The appearance of isosbestic points confirms the conversion between free and bound peptide states.
Practical Workflow:
- Prepare 10 µM GHK in deionized water.
- Add CuCl₂ incrementally while maintaining pH ≈ 7.4.
- Measure spectra from 200–800 nm after each addition.
- Plot absorbance vs. molar ratio to determine binding constant.
UV–Vis not only identifies coordination but quantifies it. This information lays the groundwork for kinetic modeling and deeper structural interpretation.
Circular Dichroism (CD) Spectroscopy
CD spectroscopy monitors conformational changes in peptides upon metal binding. It distinguishes between secondary structures like α-helices or β-turns, which can shift upon copper association.
When GHK binds Cu²⁺, characteristic negative bands appear in the far-UV region (190–250 nm), signifying new folding patterns that stabilize the complex.
Researchers interpret these signals to infer backbone rigidity, intramolecular hydrogen bonding, and the degree of coordination-induced structuring.
Advantages:
- Non-destructive measurement
- Rapid data acquisition
- Direct insight into folding behavior
CD complements UV–Vis by revealing how binding affects overall peptide architecture.
Electron Paramagnetic Resonance (EPR) Spectroscopy
EPR spectroscopy is uniquely suited for detecting unpaired electrons in paramagnetic metal centers like Cu²⁺. It provides the most direct evidence of copper’s coordination geometry and ligand environment.
EPR spectra of GHK-Cu typically display axial symmetry with g∥ ≈ 2.20 and A∥ ≈ 190 × 10⁻⁴ cm⁻¹, consistent with square-planar or elongated octahedral geometry.
By comparing g and A values, researchers confirm ligand types and bonding strength.
EPR Data Interpretation Workflow:
- Freeze GHK-Cu sample at 77 K to capture stable spectra.
- Record at X-band frequencies (~9 GHz).
- Fit hyperfine splitting constants using simulation software.
- Correlate results with UV–Vis findings for cross-validation.
EPR thus bridges electronic structure with kinetic and thermodynamic data.
Fourier Transform Infrared (FTIR) Spectroscopy
FTIR spectroscopy identifies functional groups contributing to copper coordination. Amide I and II bands (1,650 cm⁻¹ and 1,550 cm⁻¹) shift upon copper binding, indicating interaction with peptide carbonyl and amine groups.
Spectral deconvolution reveals specific residues participating in coordination, further refining Copper peptide analysis.
Key Takeaway
Combined spectroscopic techniques offer a full picture — UV–Vis for quantification, CD for conformation, EPR for geometry, and FTIR for bonding detail.
Chromatographic Techniques for GHK-Cu Characterization
While spectroscopy elucidates structure, chromatography separates, purifies, and quantifies complex mixtures, ensuring analytical accuracy.
High-Performance Liquid Chromatography (HPLC)
HPLC remains the benchmark method for verifying GHK-Cu purity and composition. Using reversed-phase (RP) columns, researchers separate the free peptide from the copper-bound form based on hydrophobicity differences.
Step-by-Step Analytical Setup:
- Column: C18, 250 mm × 4.6 mm, 5 µm particles.
- Mobile phase: water + 0.1% TFA (A) / acetonitrile + 0.1% TFA (B).
- Gradient: 5–50% B over 20 minutes.
- Detection: 220 nm (peptide) and 650 nm (Cu²⁺ complex).
The retention time shift upon metal binding quantifies how polarity changes. Peak integration gives direct concentration data, while purity percentages confirm preparation consistency.
HPLC’s reproducibility and sensitivity make it indispensable for quality assurance in Copper peptide analysis.
Liquid Chromatography–Mass Spectrometry (LC–MS)
LC–MS integrates separation with molecular identification. The technique detects intact complexes and fragmentation pathways, providing a deeper understanding of binding sites.
In GHK-Cu studies, the parent ion peak appears near m/z ≈ 404. Fragmentation reveals daughter ions representing partially coordinated fragments, confirming which atoms donate electrons to the copper center.
Analytical Insight:
- Confirms 1:1 peptide-to-metal ratio.
- Identifies side reactions or degradation products.
- Enables isotope pattern analysis for precision confirmation.
LC–MS data validate conclusions drawn from spectroscopic evidence, creating a unified analytical profile.
Inductively Coupled Plasma–Mass Spectrometry (ICP–MS)
For quantifying trace copper content, ICP–MS provides unrivaled sensitivity down to parts-per-trillion. Researchers use it to determine absolute copper concentration after chromatographic separation.
By correlating ICP–MS and LC–MS data, laboratories verify mass balance, ensuring accurate interpretation of copper-binding kinetics.
Key Takeaway
Chromatography ensures separation and quantification precision, complementing spectroscopy’s structural depth.
Integrating Spectroscopy and Chromatography in a Unified Workflow
Why Integration Matters
Modern analytical labs rarely rely on a single technique. By combining spectroscopy and chromatography, scientists gain multidimensional data that validate each other.
For example:
- UV–Vis identifies coordination onset.
- HPLC separates complex species.
- LC–MS confirms composition.
- EPR finalizes structure interpretation.
Example Integrated Workflow
- Conduct UV–Vis titration to locate binding saturation.
- Inject aliquots into HPLC to separate unbound and bound fractions.
- Analyze each fraction via LC–MS for molecular confirmation.
- Run EPR on isolated complex to confirm coordination geometry.
- Cross-compare kinetic constants derived from all methods.
This workflow minimizes experimental uncertainty and enables predictive modeling of GHK-Cu’s behavior.
Data Cross-Validation
Spectroscopic peaks can sometimes overlap or broaden, especially at high peptide concentrations. Chromatographic separation resolves this, while MS verifies mass identity. When results from all methods align, confidence in Copper peptide analysis increases substantially.
Computational and Hybrid Analytical Models
Quantum Chemical Calculations (DFT)
Density Functional Theory (DFT) allows researchers to model the electronic structure of GHK-Cu. It calculates orbital interactions and predicts binding energies that mirror experimental findings.
Typical DFT results for GHK-Cu show strong overlap between copper d-orbitals and nitrogen lone pairs, supporting a square-planar geometry. These simulations predict spectroscopic transitions later observed in UV–Vis and EPR data.
Molecular Dynamics (MD) Simulations
MD simulations extend analysis by tracking atomic motion over time. They reveal how copper binds under varying temperatures or solvent conditions.
For instance, simulations demonstrate that copper migrates between histidine and lysine residues before reaching equilibrium. This behavior aligns with experimental copper-binding kinetics data, validating both methodologies.
Machine Learning in Analytical Chemistry
Emerging machine learning (ML) tools process thousands of spectra and chromatograms to identify subtle trends humans might miss. By training models on GHK-Cu datasets, algorithms can predict unknown spectra or detect impurities automatically.
Practical Computational Workflow Example
A practical computational workflow for Copper peptide analysis usually begins with structure optimization in software such as Gaussian, ORCA, or NWChem.
Researchers import a 3D GHK structure, define the copper center, and perform geometry optimization using a hybrid functional like B3LYP with LANL2DZ basis sets for Cu²⁺.
Once the geometry converges, frequency calculations ensure the structure represents a true energy minimum. Simulated UV–Vis and EPR spectra are then generated and compared with laboratory data. This combined approach validates experimental observations and accelerates discovery.
To visualize time-dependent changes, molecular dynamics (MD) software such as GROMACS or AMBER can simulate peptide motion in water for several nanoseconds. Snapshots are analyzed to determine how temperature and ionic strength affect copper-binding kinetics.
The outcome of such computational pipelines is reproducibility: any researcher can replicate the virtual model and compare it with experimental spectra, ensuring transparent analytical validation.
Key Takeaway
Computational models extend beyond static measurement, offering dynamic predictions that guide experimental optimization.
Practical Laboratory Strategies for Reliable Copper Peptide Analysis
Sample Preparation
- Use ultrapure reagents to avoid metal contamination.
- Adjust pH using non-chelating buffers such as HEPES or MOPS.
- Filter all solutions through 0.22 µm membranes before measurement.
These precautions prevent interference that could distort Copper peptide analysis results.
Calibration and Validation
- Prepare standard curves for both copper and peptide components.
- Run blank samples to correct for background signals.
- Validate each instrument daily for wavelength and mass accuracy.
Following these procedures guarantees data reproducibility across sessions.
Data Integrity and Documentation
Detailed logging of parameters — buffer composition, temperature, pH, and equipment models — ensures transparency. Digital laboratory notebooks make cross-study comparisons easier and support peer verification.
Key Takeaway
Accurate preparation, calibration, and documentation form the backbone of reproducible copper peptide research.
Research Implications and Emerging Trends
Expanding Analytical Horizons
As instrumentation improves, researchers can now monitor copper coordination in real time using advanced time-resolved spectroscopies. These developments reveal binding events that occur within milliseconds, enriching understanding of copper-binding kinetics.
Portable Analytical Platforms
Miniaturized HPLC and handheld spectrometers now allow on-site peptide testing, supporting decentralized research environments. Compact systems offer acceptable precision for preliminary Copper peptide analysis before detailed lab confirmation.
Sustainability in Analytical Chemistry
Modern laboratories increasingly adopt greener solvents and energy-efficient chromatography systems. Reducing acetonitrile usage and switching to water-based eluents align analytical science with sustainable practices.
Automation and Data Fusion
Integrating automated sampling with AI-driven analysis streamlines workflows. Automated chromatography injectors and real-time spectral interpretation software reduce manual error while improving throughput.
Open-Access Data and Collaborative Platforms
Open-access databases now play a crucial role in Copper peptide analysis. Repositories such as Zenodo, Figshare, and ProteomeXchange allow scientists to deposit raw spectroscopic and chromatographic data sets. This practice enables comparative studies, enhances reproducibility, and speeds peer review.
Cross-institutional collaboration has also improved analytical standards. Shared calibration files and benchmark spectra reduce discrepancies between instruments. Many laboratories follow emerging FAIR principles — Findable, Accessible, Interoperable, and Reusable — to manage analytical data responsibly.
Integration with Robotic Automation
Robotic liquid handlers are transforming sample preparation. Automated pipetting and microplate systems can process hundreds of GHK-Cu samples per day while maintaining consistent temperature and pH. This automation improves statistical significance and minimizes human error.
Future analytical facilities are expected to feature fully integrated robotic chromatography coupled with cloud-based spectral processing. Such systems will continuously refine copper-binding kinetics models with real-time data inputs.
Applications and Pathways for Future Research
Analytical Method Development
GHK-Cu serves as a model compound for optimizing spectroscopic calibration curves. Its reproducible behavior across instruments allows laboratories to benchmark new analytical systems.
Comparative Metal-Peptide Studies
By substituting copper with other metals like zinc or nickel, researchers compare coordination preferences and rate differences. Such studies clarify how specific atomic properties influence copper-binding kinetics versus other metals.
Designing Novel Peptide-Metal Complexes
Analytical insights from GHK-Cu guide the design of synthetic analogs for future coordination studies. Adjusting amino acid sequences or pH conditions helps test theoretical predictions in real-world scenarios.
For researchers working with controlled laboratory compounds, always ensure sourcing from certified suppliers. It’s best to Shop Research Peptides only from trusted laboratory-grade distributors adhering to purity standards.
Integration with Computational Pipelines
Modern labs merge analytical outputs directly with molecular modeling platforms. These data-driven systems reduce experimental redundancy and accelerate result interpretation.
Collaboration Between Academia and Industry
Joint programs between universities and analytical equipment manufacturers continue to refine instrumentation sensitivity. These collaborations yield new detectors and data analysis tools that further enhance Copper peptide analysis.
Extended Analytical Examples
Example 1 – Monitoring Copper Uptake Rate
Using stopped-flow UV–Vis spectroscopy, scientists can observe binding within milliseconds. GHK’s rapid copper uptake displays a biphasic kinetic curve, confirming both fast initial chelation and slower structural reorganization.
Example 2 – Temperature Dependence of Binding
By performing titrations at multiple temperatures, activation energy for copper association can be calculated via Arrhenius plots. This quantifies how thermal fluctuations influence copper-binding kinetics.
Example 3 – Comparative Spectroscopic Modeling
Simulated EPR spectra compared with experimental data validate theoretical models. Deviations prompt researchers to refine computational parameters, improving predictive accuracy.
10. Future Analytical Perspectives
Analytical chemistry is transitioning toward fully integrated, cloud-connected systems. Real-time data transmission from spectrometers and chromatographs allows remote experts to monitor ongoing experiments globally.
Moreover, advances in quantum computing are expected to revolutionize theoretical modeling of copper–peptide interactions, solving complex wavefunction calculations previously infeasible on classical machines.
These technologies will create new benchmarks for precision, consistency, and innovation in Copper peptide analysis.
Artificial Intelligence and Predictive Modeling
Artificial intelligence (AI) is beginning to interpret complex spectroscopic signatures automatically. Machine-learning algorithms trained on large datasets of metal–peptide spectra can predict coordination geometry before physical measurement. These systems shorten analytical cycles and flag anomalies during live experiments.
Cloud-connected AI dashboards now visualize copper-binding progress curves in real time, allowing rapid decisions on sample stability. Researchers can remotely adjust parameters such as wavelength range or gradient slope without halting a run.
Quantum and Data-Driven Futures
Quantum-level simulations promise even greater accuracy. Next-generation processors will calculate electron correlation in copper orbitals at unprecedented resolution, revealing subtle shifts in copper-binding kinetics unseen by classical methods. Combined with global data sharing and AI, this will create a self-improving analytical ecosystem — one that continuously refines theoretical and experimental harmony.
Conclusion
The analytical characterization of GHK-Cu embodies the progress of modern chemical research — a union of precision, reproducibility, and interdisciplinary integration.
Through spectroscopic methods like UV–Vis, CD, EPR, and FTIR, scientists capture molecular-level data on electronic transitions and coordination geometry. Chromatographic systems such as HPLC and LC–MS add quantitative rigor, ensuring accurate separation and verification.
Together, these analytical pillars reveal the intricate copper-binding kinetics that govern stability and reactivity in peptide-metal complexes.
As technology evolves, hybrid methodologies that merge computational modeling, automation, and machine learning will further refine accuracy and speed. This continuous innovation strengthens research quality and reliability for generations of analytical chemists.
For those seeking verified laboratory-grade compounds to explore advanced copper-peptide interactions, it is essential to Shop Research Peptides only through regulated channels ensuring purity and traceability.
For in-depth analytical resources and professional guidance on peptide-based studies, visit Innovative Men, a trusted platform for technical knowledge and research collaboration.